WhatsApp, WeChat & Telegram : +8801886229488, Email : aiopharma@outlook.com
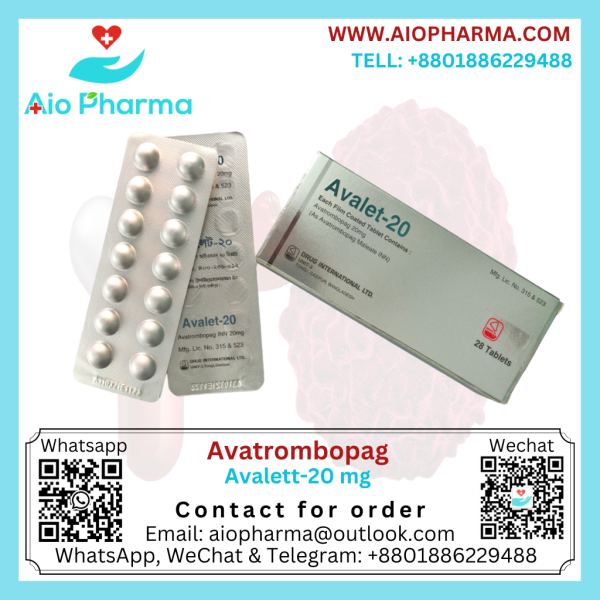
Avalet 20 mg (Avatrombopag)
Generic Name : Avatrombopag
Manufacturer : Drug International Pharma
Tablet : 28’s Strip
Originator : Doptelet by AkaRx, Inc.
Contact For Order :
WhatsApp & WeChat : +8801886229488
Email : aiopharma@outlook.com
WeChat QR Code
Avalet 20 mg (Avatrombopag) is used to treat thrombocytopenia. Order genuine Avalet 20 mg (Avatrombopag) from Aio Pharma, a trusted global supplier. We provide WHO-GMP certified products with verified cold-chain shipping for thrombocytopenia treatment. Contact us for bulk pricing and international delivery.
Why Choose Aio Pharma for Your Avalet 20 mg?
At Aio Pharma, we understand that access to genuine, life-saving medications like Avatrombopag is critical. We source our Avalet 20 mg directly from Drug International Pharma, a trusted WHO-GMP certified manufacturer in Bangladesh.
We guarantee:
-
100% Genuine Product: Every batch is verified for authenticity before shipping.
-
Secure Global Delivery: We offer discreet, temperature-controlled packaging for sensitive medicines, with tracking provided worldwide.
-
Dedicated Support: Our team is available 24/7 via WhatsApp and Telegram (+8801886229488) to assist with orders, customs documentation, and any questions.
Don’t risk counterfeit medications. Order your supply of Avalet 20 mg from a trusted partner.
How Avalet 20 mg Helps You: Avalet 20 mg (Avatrombopag) is an oral medication designed to increase low platelet counts. It is commonly prescribed for two conditions:
-
Chronic Immune Thrombocytopenia (ITP): When your immune system attacks your own platelets.
-
Chronic Liver Disease (CLD): When low platelets complicate necessary medical procedures.
Unlike injections, Avalet 20 mg is taken once daily with food, making it a convenient option for long-term management. Most patients see improved platelet counts within 1-2 weeks of starting treatment.
Avalet 20 mg works by helping your bone marrow produce more platelets. It is taken by mouth, reaches full effect in 5-6 hours, and stays in the body for about 19 hours before being naturally eliminated.
Important: Always follow your doctor’s dosage instructions. Do not stop or change your dose without medical advice.
Treating Low Platelet Counts in CLD & ITP: Treatment of Thrombocytopenia in Patients with Chronic Liver Disease (CLD): It is indicated for the treatment of thrombocytopenia in adult patients with chronic liver disease who are scheduled to undergo a procedure. For more information visit
Treatment of Thrombocytopenia in Patients with Chronic Immune Thrombocytopenia (ITP): It is indicated for the treatment of thrombocytopenia in adult patients with chronic immune thrombocytopenia who have had an insufficient response to previous treatment.
Dosage and administration: Recommended Starting Dose for Chronic ITP:
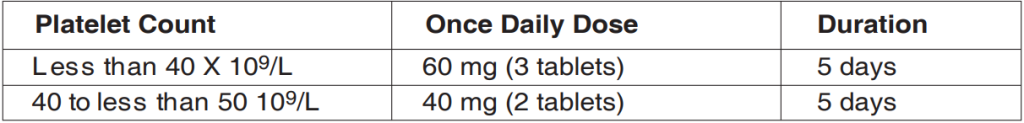
Table 1: Recommended Dose and Duration in Patients with Chronic Liver Disease Scheduled to Undergo a Procedure
Recommended Dosage for Patients With Chronic Immune Thrombocytopenia: Initial Dose Regimen: Begin Avatrombopag at a starting dose of 20mg (1tablet) once daily with food.
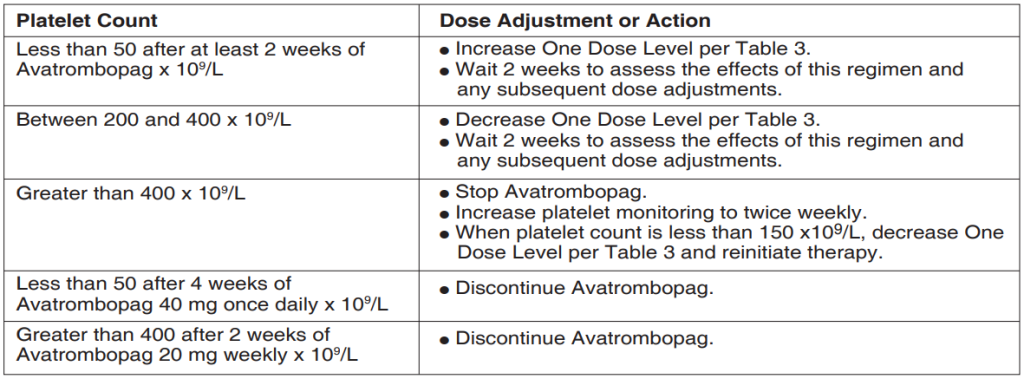
Table 2: Avatrombopag Dose Adjustments for Patients with Chronic Immune Thrombocytopenia
Table 3: Avatrombopag Dose Levels for Titration in Patients with Chronic Immune Thrombocytopenia
Precautions: Thrombotic/Thromboembolic Complications :
Avalet 20 mg is a thrombopoietin (TPO) receptor agonist and TPO receptor agonists have been associated with thrombotic and thromboembolic complications in patients with chronic liver disease or chronic immune thrombocytopenia. Monitor platelet counts and for thromboembolic events and institute treatment promptly.
Side effects:
What to expect: Most side effects are mild to moderate. They often improve as your body adjusts to the medication.
For patients with Chronic Liver Disease (CLD):
-
Very common (may affect more than 1 in 10 people): Fever, abdominal pain, nausea, headache, fatigue, swollen feet/ankles (peripheral edema)
For patients with Chronic Immune Thrombocytopenia (ITP):
-
Common: Headache, fatigue, confusion
-
Less common but possible: Nosebleeds (epistaxis), upper respiratory infection, joint pain (arthralgia), bleeding gums, small red spots on skin (petechiae), stuffy/runny nose (nasopharyngitis)
When to call your doctor: If any side effect becomes severe, does not go away, or if you notice unusual bleeding or bruising.
Use in Pregnancy and Lactation: This medication may harm an unborn baby. Do not take Avalet 20 mg if you are pregnant, planning to become pregnant, or breastfeeding.
Pregnant women should be advised of the potential risk to a fetus. Females of reproductive potential should be advised to inform their prescriber of a known or suspected pregnancy.
Nursing Mothers: Women should be advised not to breastfeed during treatment with Avatrombopag and for at least 2 weeks after the final dose.
Action required: Use effective birth control during treatment. Talk to your doctor immediately if you become pregnant.
Drug interactions: In patients starting moderate or strong dual inhibitors of CYP2C9 and CYP3A4 while receiving Avatrombopag, monitor platelet counts and adjust Avatrombopag dose as necessary.
Overdose: There is no data available.
Storage: Store below 30o C in a dry place.
Packing: Each box contains 28’s tablets in a blister pack
Disclaimer: The information provided is for general informational purposes only and is not a substitute for professional medical advice. Always consult your doctor or a qualified healthcare provider before taking any medication.








Jhon –
Good Medicine
Li yansi –
效果很好
Mike –
Highly recommended
Rick –
I’m fully satisfied. You guys doing great. Thank you