WhatsApp, WeChat & Telegram : +8801886229488, Email : aiopharma@outlook.com
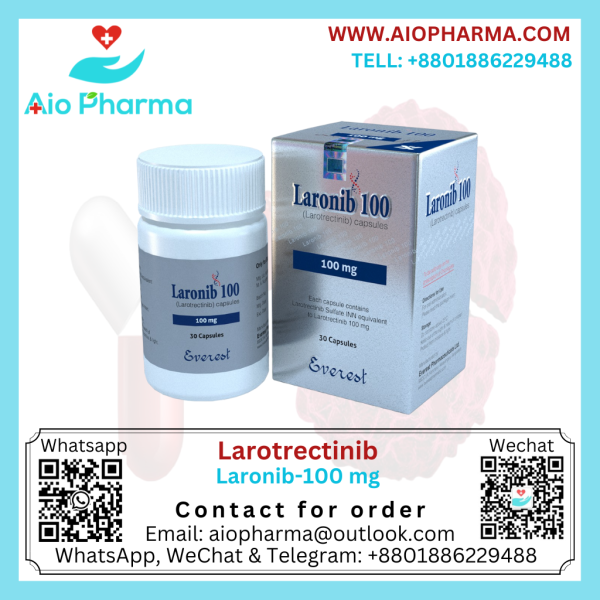
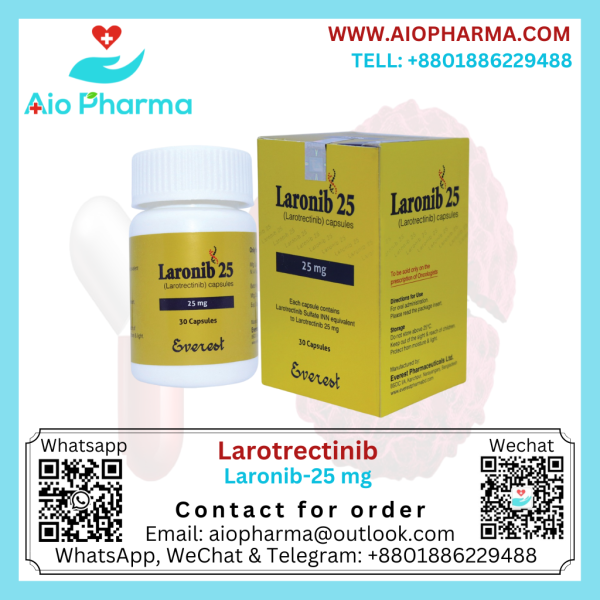
Larotrectinib (Laronib 100mg/25mg)
Generic Nam: Larotrectinib
Manufacturer: Everest Pharma
Pack Size: 30’s Pot
Originator: Vitrakvi by Array BioPharma & Bayer
Contact For Order:
WhatsApp & WeChat: +8801886229488
Email: aiopharma@outlook.com
WeChat QR Code
Laronib Larotrectinib Vitrakvi is used to treat solid tumors. It was discovered by Array BioPharma and licensed to Loxo Oncology in 2013. In Bangladesh, this medicine is manufactured by Everest Pharmaceuticals LTD.
Q: What type of drug is Larotrectinib?
It is an inhibitor of tropomyosin kinase receptors TrkA, TrkB, and TrkC. Array BioPharma discovered and licensed larotrectinib to Loxo Oncology in 2013. The FDA initially awarded orphan drug status to larotrectinib in 2015 for soft tissue sarcoma, and breakthrough therapy designation in 2016 for the treatment of metastatic solid tumors with NTRK fusion. for more visit
Q: What are the side effects of Larotrectinib?
Larotrectinib side effects. liver problems–loss of appetite, stomach pain (upper right side), nausea, vomiting, dark urine, clay-colored stools, jaundice (yellowing of the skin or eyes). doctor may delay or permanently discontinue your cancer treatments if you have certain side effects. for more visit
Q: How should I take Larotrectinib (Laronib)?
Larotrectinib comes in capsules (25 mg and 100 mg) and as an oral solution. Take larotrectinib exactly as your healthcare provider tells you. Your healthcare provider may stop treatment or change your dose of larotrectinib if you have side effects.
Q: Is Larotrectinib an immunotherapy?
Larotrectinib allows us to treat a select patient population based solely on their mutation, regardless of cancer type. Our work builds on the recent approval of pembrolizumab (Keytruda®), an immunotherapy for any tumor that harbors a specific pattern of DNA damage known as microsatellite instability.
Q: Is Larotrectinib chemotherapy?
Laronib/Larotrectinib/Vitrakvi is an oral medicine that is not chemotherapy.
Q: How effective is Larotrectinib?
In the final trial analysis, larotrectinib produced an overall response rate of 75% and entrectinib generated 57% with a sustained duration of responses in 39% and 45% of patients at 12 months, respectively (Bayer Healthcare Pharmaceuticals, Inc., 2019; Genentech USA, Inc., 2019).







Jacinto –
deliver on time