WhatsApp, WeChat & Telegram : +8801886229488, Email : aiopharma@outlook.com
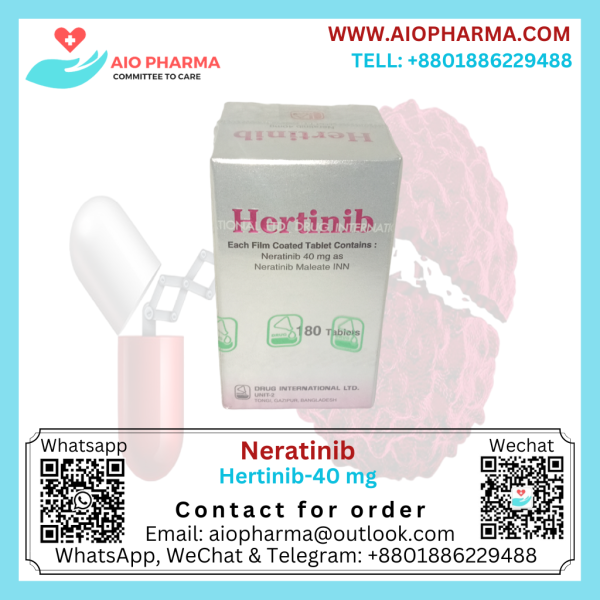
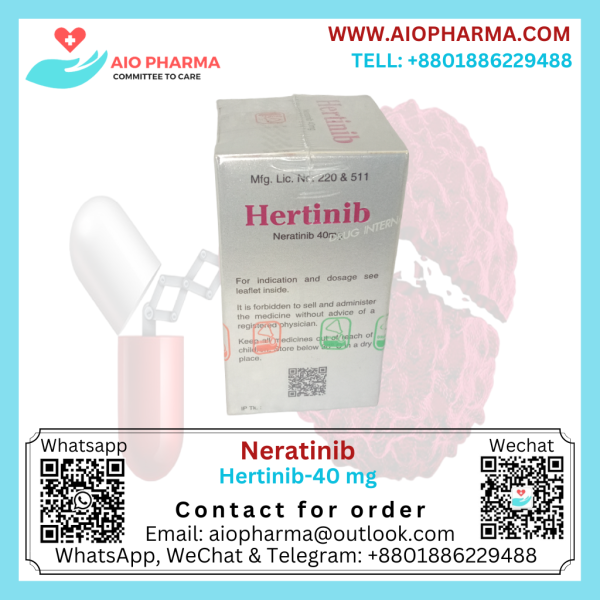
Neratinib 40 mg Hertinib
Generic Name: Neratinib
Manufacturer: Drug INT
Contact For Order:
WhatsApp & WeChat: +8801886229488
Email: aiopharma@outlook.com
WeChat QR Code
Neratinib 40 mg Hertinib is used to treat breast cancer. Neratinib is given after you have surgery and have completed treatment with trastuzumab.
FAQ About Neratinib 40 mg Hertinib
Q: What type of drug is Neratinib 40 mg Hertinib?
Neratinib is in a class of medications called kinase inhibitors. It works by blocking the action of an abnormal protein that signals cancer cells to multiply.
Q: What is neratinib used for?
Nerlynx (chemical name: neratinib) is a type of targeted therapy called an irreversible pan-HER inhibitor used to treat HER2-positive breast cancer. Nerlynx is used to treat early-stage HER2-positive breast cancer for an extended period after surgery (doctors call this extended adjuvant therapy). Source
Q: Is neratinib a chemotherapy drug?
Neratinib is considered a “targeted therapy” medication, not a chemotherapy drug. To date, cancer treatment has focused primarily on killing rapidly dividing cells. This is because a major feature of cancer cells is that they divide rapidly.
Q: Does neratinib cross the blood-brain barrier?
Neratinib as an oral medication had demonstrated success against HER2+ type of cancer as the maleic acid salt. Due to its molecule size, structure, and solubility, Neratinib maleate could not sufficiently cross the blood-brain barrier (BBB) to reach those affected areas even with a log P of 3.16. Source
Dose Modifications: For Adverse Reactions: Hertinib dose modification is recommended based on individual safety and tolerability. Hertinib should be discontinued for patients who fail to recover to Grade 0-1 from treatment-related toxicity, for toxicities that result in a treatment delay of> 3 weeks, or for patients that are unable to tolerate 120 mg daily. Additional clinical situations may result in dose adjustments as clinically indicated (e.g. intolerable toxicities, persistent Grade 2 adverse reactions, etc.).
Side Effects:
- Diarrhea
- Hepatotoxicity
Advanced or Metastatic Breast Cancer: Hertinib in combination with Capecitabine is indicated for the treatment of adult patients with advanced or metastatic HER2-positive breast cancer who have received two or more prior anti-HER2-based regimens in the metastatic setting.
Dosage and Administration: Antidiarrheal Prophylaxis: Antidiarrheal prophylaxis is recommended during the first 2 cycles (56 days) of treatment and should be initiated with the first dose of Hertinib. Additional antidiarrheal agents may be required to manage diarrhea in patients with loperamide-refractory diarrhea. Hertinib dose interruptions and dose reductions may also be required to manage diarrhea. The recommended dose of Hertinib is 240 mg (six tablets) given orally once daily with food, continuously for one year. Patients should be instructed to take Hertinib at approximately the same time every day. Hertinib tablets should be swallowed whole (tablets should not be chewed, crushed, or split before swallowing). If a patient misses a dose, the missed dose should not be replaced, and patients should be instructed to resume Hertinib with the next scheduled daily dose. Or, as directed by the registered physicians.
Use in Pregnancy and Lactation: Hertinib can cause fetal harm when administered to a pregnant woman. There are no available data on pregnant women to inform the drug-associated risk. Pregnant women should be advised of the potential risk to a fetus.
Packing: Each container contains 180 tablets in a box.
How to order?
Ordering from aiopharma.com is easy and convenient. While we don’t currently have an automatic order system on our website, we offer several options for placing an order:
- WhatsApp: You can send us a message on WhatsApp at +8801886229488 and one of our customer service representatives will assist you in placing your order.
- WeChat: We also accept orders through WeChat. Simply add our WeChat number +8801886229488 and send us a message to get started.
- Messenger: If you prefer to use Facebook Messenger, you can send us a message and we’ll be happy to help you place your order.
- Email: You can also send us an email at aiopharma01@gmail.com or aiopharma@outlook.com with your order details, and we’ll get back to you as soon as possible. or contact us
No matter which method you choose, our team is dedicated to providing you with personalized service and ensuring a smooth ordering process. If you have any questions or concerns, please don’t hesitate to contact us.
What are the payment methods available?
At aiopharma.com, we offer several payment options to make it easy and convenient for you to purchase our high-quality products. However, please note that our website does not have a payment system, so payment must be made manually like ordering. Here are the payment methods we accept:
- Western Union
- MoneyGram
- Ria Transfer
- Bank Transfer
Once you have placed your order through one of our available methods (WhatsApp, WeChat, Messenger, or email), we will provide you with instructions for making payment via your preferred payment method. Once payment has been received, we will process and ship your order as soon as possible. Know about aio pharma at luckycapsules.com
If you have any questions or concerns about our payment options, please don’t hesitate to contact us. Our team is always available to assist you.







Reviews
There are no reviews yet.